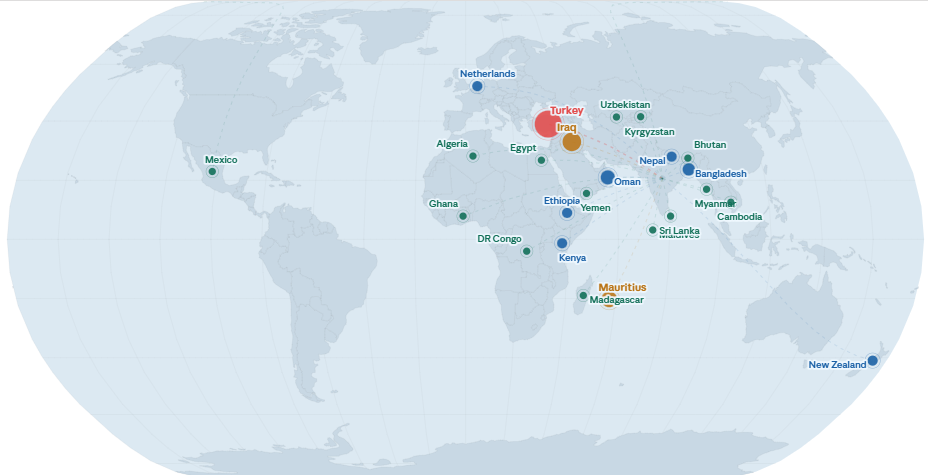
A patient from Mauritius with relapsed aggressive non-Hodgkin’s lymphoma travelled to India for an indigenously manufactured CAR-T cell therapy by ImmunoACT, a treatment that would have cost millions of dollars in the United States. His story is part of a quiet but growing trend: ImmunoACT has now treated close to 60 patients from 20+ countries across five continents positioning India as an emerging destination for accessible CAR-T therapy [based on data collected from company and treatment centres].
When Mr. Jeetah’s oncologist in Mauritius told him there was nothing more they could do for his relapsed diffuse large B-cell lymphoma, he did what many patients in his position do — he looked outward. He identified a treatment option in Mumbai with a different cost structure compared to what had been quoted in other regions.
“I couldn’t afford a western CAR-T therapy,” he says. “We’re talking millions of dollars. I don’t even have assets to sell to get that kind of money. My treatment in India got funded by my pension fund.”
The Global Context: CAR-T as a Therapy Few Can Afford
CAR-T therapy — chimeric antigen receptor T-cell therapy — has transformed outcomes for certain patients with relapsed or refractory B-cell blood cancers. It involves extracting a patient’s own immune T-cells, genetically engineering them to identify specific receptors on cells on certain types of cancer, and reinfusing them after laboratory expansion.
But access has remained a critical barrier. For patients outside high-income countries with robust insurance systems, this effectively places the therapy out of reach.
India’s emergence as a CAR-T destination is changing that equation.
An Indigenous Therapy Built in India, for the World
ImmunoACT’s CAR-T therapy, developed in partnership with IIT Bombay and approved by India’s Central Drugs Standard Control Organisation (CDSCO) in 2023, is the country’s first domestically developed and manufactured CAR-T treatment. Unlike imported therapies that require complex international cell shipment, this therapy is produced entirely within India.
The product also holds a US patent for a humanised CAR construct; a design element aligned with immunologic engineering principles seen in next-generation cellular therapies globally. Since launch, it has been administered to more than 650 patients with relapsed or refractory B-cell malignancies across India, according to company disclosures. [internal data; detailed evidence available upon request].
Internationally, ImmunoACT has now treated patients from 24 countries spanning five continents — from Turkey, Iraq, and Oman across the Middle East, to Kenya, Ethiopia, and the Democratic Republic of Congo in Africa, to as far as New Zealand and Mexico. That geographic reach — built entirely through clinical outcomes and physician referrals, not through multinational distribution — reflects increasing international patient utilization.
According to Dr. Kunal Goyal, its administration requires specialised clinical expertise. He emphasized that patient outcomes and safety profiles may vary, and that timely recognition and management of adverse events are critical.
Cost, Transparency, and the Patient’s Decision
“Dr. Goyal laid out the full cost in detail. He told me it cannot get more expensive than this unless there are very severe side effects requiring costly medication. And in practice, the cost was way lower than what other south Asian countries had quoted.” For patients navigating a system they do not know in a country they have never visited, that kind of directness is, he says, its own form of care.
India’s domestic manufacturing and relatively independent supply chain model has enabled differentiated pricing that is very difficult to replicate with imported therapies.
India’s Growing Role in Global Cancer Care
India has long attracted international patients for cardiac surgery, orthopaedics, and transplant medicine. CAR-T therapy represents a new frontier — and a more complex one. With over 90 active collection and infusion sites across India and a manufacturing base that eliminates the need for overseas cell logistics, the ecosystem is expanding to support international patients at scale.
The diversity of countries now represented in ImmunoACT’s international patient base — ranging from high-income nations in Europe and Oceania to emerging economies across Africa and Central Asia — suggests that the draw is not purely economic.
For patients like Mr. Jeetah — those who have relapsed after standard treatment, for whom transplant is unlikely to succeed, and for whom the cost of Western therapy is simply not a real option — India can slowly become a preferred choice in future. More than a year and half into remission, he remains under regular follow-up — one of 60 international patients who crossed borders for a chance at life and found it in India.
Note: CAR-T therapy may cause side effects that are severe or life-threatening, requiring intensive monitoring and specialised hospital care. Treatment outcomes vary based on disease characteristics, prior therapies, and individual patient factors. Patients considering CAR-T therapy are advised to discuss the full risk-benefit profile with their treating physician. This article is written for knowledge and educational purposes and does not constitute medical advice.